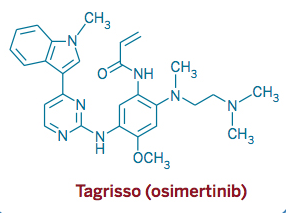
AZD 9291, Osimertinib
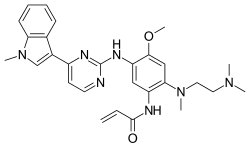
2-Propenamide, N-[2-[[2-(dimethylamino)ethyl]methylamino]-4-methoxy-5-[[4-(1-methyl-1H-indol-3-yl)-2-pyrimidinyl]amino]phenyl]-
N-(2-((2-(dimethylamino)ethyl)(methyl)amino)-4-methoxy-5-((4-(1-methyl-1H-indol-3-yl)pyrimidin-2-yl)amino)phenyl)acrylamide.
cas :1421373-65-0, 1421373-66-1(mesylate salt)
cas :1421373-65-0, 1421373-66-1(mesylate salt)
UPDATE............FDA APPROVED NOV2015
EU ...... 3 FEB 2016 APPROVED
03 February 2016
AstraZeneca today announced that the European Commission (EC) has granted conditional marketing authorisation for TAGRISSO™ (AZD9291, osimertinib) 80mg once-daily tablets for the treatment of adult patients with locally advanced or metastatic epidermal growth factor receptor (EGFR) T790M mutation-positive non-small cell lung cancer (NSCLC).
Osimertinib is indicated for patients with T790M mutation-positive NSCLC, irrespective of previous treatment with a
ASTRAZENECA

Mechanism of Action: Third-generation, oral, irreversible, selective epidermal growth factor receptor (EGFR) inhibitor
Non-small cell lung cancer (NSCLC)
AZD-9291 M. Wt: 499.61
AZD-9291 Formula: C28H33N7O2
AZD-9291 Formula: C28H33N7O2
AZD9291, a third-generation orally irreversible epidermal growth factor receptor (EGFR) inhibitor, is under development by British drug maker AstraZeneca for the treatment of patients with metastatic EGFR T790M mutation-positive non-small cell lung cancer (NSCLC).
Lung cancer is the major cause of cancer death in the world while non small cell lung cancer (NSCLC) accounts approx. 85% of all lung cancer diagnosis. Approximately 50% of non–small cell lung cancer (NSCLC) patients who develop resistance to inhibitors of the epidermal growth factor receptor (EGFR) have acquired a second mutation, T790M. There are currently no approved treatments for patients who develop a T790 mutation.
Lung cancer is the major cause of cancer death in the world while non small cell lung cancer (NSCLC) accounts approx. 85% of all lung cancer diagnosis. Approximately 50% of non–small cell lung cancer (NSCLC) patients who develop resistance to inhibitors of the epidermal growth factor receptor (EGFR) have acquired a second mutation, T790M. There are currently no approved treatments for patients who develop a T790 mutation.

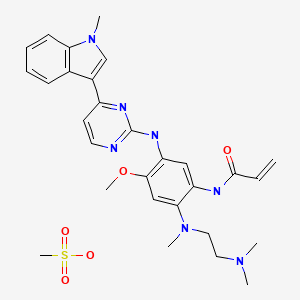
AZD9291 mesylate
Also known as: AZD 9291 mesylate; AZD-9291 mesylate; HY-15772A; 1421373-66-1
AZD-9291 is a potent and selective mutated forms EGFR inhibitor(Exon 19 deletion EGFR IC50=12.92 nM, L858R/T790M EGFR IC50= 11.44 nM, wild type EGFR IC50= 493.8 nM).

Osimertinib (previously known as mereletinib[2] and AZD9291; trade name Tagrisso) is a third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) drug[3][4] developed by AstraZeneca Pharmaceuticals - for mutated EGFR cancers.

Approvals and indications
In November 2015, after a Priority Review, the US FDA granted accelerated approval to osimertinib for the treatment of metastaticepidermal growth factor receptor (EGFR) T790M mutation-positive non-small cell lung cancer (NSCLC), as detected by an FDA-approved test, which has progressed on or after EGFR tyrosine kinase inhibitor (TKI) therapy.[5][6]
The FDA approval made reference to two clinical trials, in which an EGFR T790M mutation was confirmed by a Cobas EGFR mutation test; osimertinib was given as 80 mg once daily.[1][7]
AZD-9291 is a third-generation EGFR inhibitor, showed promise in preclinical studies and provides hope for patients with advanced lung cancers that have become resistant to existing EGFR inhibitors. AZD9291 is highly active in preclinical models and is well tolerated in animal models. It inhibits both activating and resistant EGFR mutations while sparing the normal form of EGFR that is present in normal skin and gut cells, thereby reducing the side effects encountered with currently available medicines

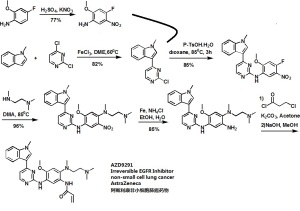
Synthesis of AZD9291,
CLICK ON IMAGES FOR CLEAR VIEW
WO 2013014448
Example 27: V-f5-{[5-Cvano-4-flH-indol-3-yl)pyrimidin-2-yllamino}-4-methoxy-2-{4- methylpiperazin-l-yl}phenyl)prop-2-enamide
Acryloyl chloride (0.100 mL, 1M in THF, 0.1 mmol) was added dropwise to a fine slurry of 2- {[5-amino-2-methoxy-4-(4-methylpiperazin- 1 -yl)phenyl] amino} -4-(lH-indol-3- yl)pyrimidine-5-carbonitrile (Intermediate 99, 47 mg, 0.10 mmol) and DIPEA (0.027 mL, 0.16 mmol) in THF (2 mL) at -10°C over a period of 2 minutes under N2.The mixture was then stirred at 0°C for 10 minutes then allowed to warm to r.t. over 20 minutes. The mixture was then cooled again to -10°C and further acryloyl chloride (0.06 mL, 1M in THF, 0.06 mmol) was added dropwise. The mixture was stirred at 0°C for a further 10 minutes, then allowed to warm to r.t. over 20 minutes. The mixture was then concentrated in vacuo and the resulting reside was dissolved in CH2C12 (2 mL). This solution was washed with sat. NaHC03 (1 mL), dried (MgS04) and concentrated in vacuo. Purification by FCC, eluting with 1.5-7% 7N methanolic ammonia in CH2C12 gave a residue that was washed with CH3OH (0.1 mL) and dried in air to give the title compound (1 lmg, 20%) as a cream crystalline solid; 1H NMR: 2.28 (3H, s), 2.54-2.65 (4H, m), 2.93 (4H, s), 3.75 (3H, s), 5.71 (1H, d), 6.18 (1H, d), 6.64 (1H, dd), 6.91 (2H, m), 7.18 (1H, s), 7.47 (1H, d), 8.02 (1H, s), 8.52 (1H, s), 8.67 (1H, s), 9.04 (1H, s), 9.40 (1H, s), 11.99 (1H, s); m/z: ES+ MH+ 509. Example 28: V-f2-{2-Dimethylaminoethyl-methylamino}-4-methoxy-5-{[4-fl- methylindol-3-yl)pyrimidin-2-yllamino}phenyl)prop-2-enamide
A solution of acryloyl chloride (34.5 mg, 0.38 mmol) in CH2C12 (1 mL) was added dropwise to a stirred mixture of N;-(2-dimethylaminoethyl)-5-methoxy-N;-methyl-N¥-[4- (l-methylindol-3-yl)pyrimidin-2-yl]benzene-l,2,4-triamine (Intermediate 100, 170 mg, 0.38 mmol) and DIPEA (0.073 mL, 0.42 mmol) in CH2C12 (5 mL), which was cooled in an ice/water bath. The mixture was stirred for 1.5h and then diluted with CH2C12 (25 mL) and washed with sat.NaHCOs (50 mL). The aqueous washes were extracted with CH2C12 (2 x 25 mL). The combined organic solutions were dried (MgSC^) and concentrated in vacuo. Purification by FCC, eluting with 0-4% 7N methanolic ammonia in CH2C12 gave the title compound (75 mg, 39%) as a cream solid after trituration with diethyl ether; 1H NMR: 2.21 (6H, s), 2.29 (2H, t), 2.72 (3H, s), 2.89 (2H, t), 3.86 (3H, s), 3.92 (3H, s), 5.77 (1H, dd), 6.27 (1H, dd), 6.43 (1H, dd), 7.04 (1H, s), 7.15 (1H, t), 7.20-7.27 (2H, m), 7.53 (1H, d), 7.91 (1H, s), 8.24 (1H, d), 8.33 (1H, d), 8.68 (1H, s), 9.14 (1H, s), 10.22 (1H, s); m/z: ES+ MH+ 500.42.
Example 28 (Alternative synthesis 1): V-f2-{2-Dimethylaminoethyl-methylamino}-4- methoxy-5-{[4-(l-methylindol-3-yl)pyrimidin-2-yllamino}phenyl)prop-2-enamide
To a stirred solution of 3-chloro-N-[2-[2-dimethylaminoethyl(methyl)amino]-4-methoxy- 5-[[4-(l-methylindol-3-yl)pyrimidin-2-yl]amino]phenyl]propanamide (Intermediate 174, 31.5 g, 58.76 mmol) in acetonitrile (310 mL) was added triethylamine (17.84 g, 176.28 mmol) at r.t. The resulting mixture was heated to 80°C for 6h then cooled to r.t.. Water (130 mL) was then added and the mixture stirred for 12h. The mixture was then filtered, washed with a mixture of water and acetonitrile (160 mL, 1 : 1) and dried at 50°C for overnight to give the title compound (19.2 g, 94%) as a solid form identified herein as polymorphic form D. 1H NMR: 2.69 (3H, s), 2.83 (6H, d), 3.35 (4H, s), 3.84 (3H, s), 3.91 (3H, s), 5.75 (IH, d), 6.28 (IH, d), 6.67 (IH, dd), 7.05-7.23 (2H, m), 7.29 (IH, t), 7.43 (IH, d), 7.56 (IH, d), 8.21 (2H, s), 8.81 (IH, s), 9.47 (IH, s), 9.52 (IH, s), m/z: ES+ MH+ 500.26.
Example 28 (Alternative synthesis 2): V-f2-{2-Dimethylaminoethyl-methylamino}-4- methoxy-5-{[4-(l-methylindol-3-yl)pyrimidin-2-yllamino}phenyl)prop-2-enamide
To a stirred solution of N1-(2-dimethylaminoethyl)-5-methoxy-N1-methyl-N4-[4-(l- methylindol-3-yl)pyrimidin-2-yl]benzene-l,2,4-triamine (Intermediate 100, 10 g, 21.32 mmol) in THF (95 mL) and water (9.5 mL) at 0°C was added the 3-chloropropanoyl chloride (3.28 g, 25.59 mmol). The mixture was stirred at r.t. for 15 minutes then NaOH (3.48 g, 85.28 mmol) was added. The resulting mixture was heated to 65°C for lOh. The mixture was then cooled to r.t. and CH3OH (40 mL) and water (70 mL) were added. The resulting mixture was stirred overnight. The resulting solid was collected by filtration, washed with water (25 mL) and dried at 50°C for 12h to give the title compound (7.0 g, 94%) as a solid form identified herein as polymorphic Form D.1H NMR: 2.69 (3H, s) 2.83 (6H, d) 3.35 (4H, s) 3.84 (3H, s) 3.91 (3H, s) 5.75 (IH, d) 6.28 (IH, d) 6.67 (IH, dd) 7.05-7.23 (2H, m) 7.29 (IH, t) 7.43 (IH, d) 7.56 (IH, d) 8.21 (2H, s) 8.81 (IH, s) 9.47 (IH, s) 9.52 (IH, s) ES+ MH+ 500.26.
Example 28A: V-f2-{2-Dimethylaminoethyl-methylamino}-4-methoxy-5-{[4-fl- methylindol-3-yl)pyrimidin-2-yll amino}phenyl)prop-2-enamide mesylate salt
AZD 9291
Procedure 1: To a stirred solution of N-[2-[2-dimethylaminoethyl(methyl)amino]-4- methoxy-5 - [[4-( 1 -methylindol-3 -yl)pyrimidin-2-yl] amino]phenyl]prop-2-enamide
(Example 28, 20 g, 36.63 mmol) in ethanol (120 mL) and EtOAc (80 mL) at 70°C was added methane sulfonic acid (3.59 g, 36.63 mmol) as a solution in EtOAc (40 mL). The resulting mixture was stirred for 1.5h. The resulting solid was collected by filtration and dried at 80°C under vacuum overnight to give the title salt (20.5 g, 94%) in a solid form defined herein as polymorphic Form B for this salt.
Procedure 2: To a stirred solution of N-[2-[2-dimethylaminoethyl(methyl)amino]-4- methoxy-5 - [[4-( 1 -methylindol-3 -yl)pyrimidin-2-yl] amino]phenyl]prop-2-enamide
(Example 28, 5 g, 9.11 mmol) in acetone (45.5 mL) and water (4.55 mL) at 50°C was added methane sulfonic acid (0.893 g, 9.11 mmol) as a solution in acetone (4.55 mL). The resulting mixture was stirred for 1.5h. The resulting solid was collected by filtration and dried at 80°C under vacuum overnight to give the title salt (4.9 g, 94%) in a solid form defined herein as polymorphic Form B for this salt; ΧΗ NMR (acetone-ii6): 2.72 (3H, s), 2.96 (3H, s), 3.01 (6H, s), 3.58 (3H, t), 3.87-3.90 (7H, m), 5.76 (1H, dd), 6.38-6.53 (2H, m), 7.12 (1H, t), 7.20 (1H, t), 7.29 (1H, s), 7.40 (2H, t), 8.07-8.16 (3H, m), 8.56 (1H, s), 9.30 (1H, s), 9.60 (1H, s), 9.66 (1H, s ); m/z: ES+ MH+ 500.26.
Procedure 3: Polymorphic Form A of N-(2-{2-dimethylaminoethyl-methylamino}-4- methoxy-5 - { [4-( 1 -methylindol-3 -yl)pyrimidin-2-yl] amino } phenyl)prop-2-enamide mesylate salt was prepared in a similar manner as described above on a ~50 mg scale, except that acetonitrile was used as the solvent. Specifically, ~9.6mg methanesulfonic acid was dissolved into a minimum volumn of acetonitrile. ~50 mg N-(2- {2-dimethylamino- ethyl-methylamino } -4-methoxy-5 - { [4-( 1 -methylindol-3 -yl)pyrimidin-2-yl] amino } phenyl)- prop-2-enamide was also dissolved into a minimum volume of acetonitrile and then the resulting solution was added to the methanesulfonic acid solution. Formation of a solid resulted upon addition. This solid was collected by filtration and was air-dried and then analysed. The particular solid form produced in this experiment was designated as
Polymorphic Form A for this salt.
 | |
| Systematic (IUPAC) name | |
|---|---|
| N-(2-{2-dimethylaminoethyl-methylamino}-4-methoxy-5-{[4-(1-methylindol-3-yl)pyrimidin-2-yl]amino}phenyl)prop-2-enamide | |
| Clinical data | |
| Trade names | Tagrisso |
| Routes of administration | Oral tablets |
| Pharmacokinetic data | |
| Protein binding | probably high[1] |
| Metabolism | oxidation (CYP3A) |
| Biological half-life | 48 hours |
| Excretion | feces (68%), urine (14%) |
| Identifiers | |
| CAS Number | 1421373-65-0 |
| PubChem | CID 71496458 |
| ChemSpider | 31042598 |
| UNII | 3C06JJ0Z2O |
| ChEBI | CHEBI:90943 |
| Chemical data | |
| Formula | C28H33N7O2 |
| Molar mass | 499.6 g/mol |
References
- "Tagrisso (osimertinib) Tablet, for Oral Use. Full Prescribing Information" (PDF). AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850. Retrieved 16 November 2015.
- "Proposed INN: List 113" (PDF). International Nonproprietary Names for Pharmaceutical Substances (INN) 29 (2): 285. 2015. Retrieved16 November 2015.
- Ayeni D, Politi K, Goldberg SB (2015). "Emerging Agents and New Mutations in EGFR-Mutant Lung Cancer". Clin. Cancer Res. 21 (17): 3818–20. doi:10.1158/1078-0432.CCR-15-1211. PMID 26169963.
- Tan CS, Gilligan D, Pacey S (2015). "Treatment approaches for EGFR-inhibitor-resistant patients with non-small-cell lung cancer". Lancet Oncol. 16 (9): e447–59. doi:10.1016/S1470-2045(15)00246-6. PMID 26370354.
- U.S. Food and Drug Administration. Hematology/Oncology (Cancer) Approvals & Safety Notifications. [1] Xu M, Xie Y, Ni S, Liu H (2015). "The latest therapeutic strategies after resistance to first generation epidermal growth factor receptor tyrosine kinase inhibitors (EGFR TKIs) in patients with non-small cell lung cancer (NSCLC)". Ann Transl Med 3 (7): 96.doi:10.3978/j.issn.2305-5839.2015.03.60. PMC 4430733. PMID 26015938.
- U.S. Food and Drug Administration. "Osimertinib". [2]

AstraZeneca R&D Facility, Alderley Park, Cheshire, United Kingdom
Cheshire Map Showing Location of Alderley Park
District: Macclesfield
Easting: 384439 Northing: 374467
Latitude: 53.27 Longitude: -2.23
 = Alderley Park
= Alderley Park
Easting: 384439 Northing: 374467
Latitude: 53.27 Longitude: -2.23
 = Alderley Park
= Alderley Park| INN or syn | URL | CID | InChIKey |
| AZ11657312 | |||
| AZ876 | AZ12260493 | CID 11655079 | IVANYIPLGFVBGR-UHFFFAOYSA-N |
| AZ12419304 | |||
| AZ12609721 | |||
| AZ12823138 | |||
| AZ12971554 | CID 44537923 | GMKHQRCPNMGCIX-ZWKOTPCHSA-N | |
| AZ13483342 | |||
| AZD0328 | CID 9794392 | OCKIPDMKGPYYJS-ZDUSSCGKSA-N | |
| Saracatinib | AZD0530 | CID 10302451 | OUKYUETWWIPKQR-UHFFFAOYSA-N |
| Barasertib | AZD1152 | CID 11497983 | GBJVVSCPOBPEIT-UHFFFAOYSA-N |
| AZD1208 | CID 58423153 | MCUJKPPARUPFJM-UWCCDQBKSA-N | |
| AZD1236 | |||
| AZD1332 | CID 49831044 | LBVKEEFIPBQIMD-UHFFFAOYSA-N | |
| AZD1480 | CID 16659841 | PDOQBOJDRPLBQU-QMMMGPOBSA-N | |
| AZD1656 | CID 16039797 | FJEJHJINOKKDCW-INIZCTEOSA-N | |
| MK175 | AZD1775 | CID 24856436 | BKWJAKQVGHWELA-UHFFFAOYSA-N |
| AZD1981 | CID 11292191 | JWYIGNODXSRKGP-UHFFFAOYSA-N | |
| AZD2014 | CID 25262792 | JUSFANSTBFGBAF-IRXDYDNUSA-N | |
| Olaparib | AZD2281 | CID 23725625 | FDLYAMZZIXQODN-UHFFFAOYSA-N |
| AZD2624 | CID 23649245 | QYTBBBAHNIWFOD-NRFANRHFSA-N | |
| AZD2927 | CID 57345449 | GAHPWXLXWUVMIV-MRXNPFEDSA-N | |
| Lesogaberan | AZD3355 | CID 9833984 | WVTGPBOMAQLPCP-GSVOUGTGSA-O |
| AZD3463 | CID 56599293 | GCYIGMXOIWJGBU-UHFFFAOYSA-N | |
| AZD3857 | |||
| AZD4017 | CID 24946280 | NCDZABJPWMBMIQ-INIZCTEOSA-N | |
| AZD4320 | |||
| AZD4547 | CID 51039095 | NCDZABJPWMBMIQ-INIZCTEOSA-N | |
| AZD4877 | CID 10368812 | SMFXSYMLJDHGIE-UHFFFAOYSA-N | |
| AZD5363 | CID 25227436 | JDUBGYFRJFOXQC-KRWDZBQOSA-N | |
| AZD5582 | CID 49847690 | WLMCRYCCYXHPQF-ZVMUOSSASA-N | |
| TX4 | AZD5904 | CID 10264211 | RSPDBEVKURKEII-ZCFIWIBFSA-N |
| Selumetinib | AZD6244 | CID 10127622 | IAYGCINLNONXHY-LBPRGKRZSA-N |
| AZD6495 | |||
| AZD6738 | |||
| Lanicemine | AZD6765 | CID 3038485 | FWUQWDCOOWEXRY-UHFFFAOYSA-N |
| AZD7325 | CID 23581869 | KYDURMHFWXCKMW-UHFFFAOYSA-N | |
| AZD7762 | CID 11152667 | IAYGCINLNONXHY-LBPRGKRZSA-N | |
| AZD8055 | CID 25262965 | KVLFRAWTRWDEDF-IRXDYDNUSA-N | |
| AZD8186 | CID 52913929 | LMJFJIDLEAWOQJ-UHFFFAOYSA-N | |
| AZD8329 | CID 25006684 | XWBXJBSVYVJAMZ-UHFFFAOYSA-N | |
| AZD8529 | CID 25125217 | IPCYZQQFECEHLI-UHFFFAOYSA-N | |
| AZD8542 | CID 53344810 | SMQVBAGSZVHCJP-UHFFFAOYSA-N | |
| AZD8931 | CID 11488320 | DFJSJLGUIXFDJP-UHFFFAOYSA-N | |
| STAT3Rx | AZD9150 | ||
| AZD9291 | CID 71496458 | DUYJMQONPNNFPI-UHFFFAOYSA-N | |
| Alvelestat | AZD9668 | CID 46861623 | QNQZWEGMKJBHEM-UHFFFAOYSA-N |
| Zibotentan | ZD4054 | CID 9910224 | FJHHZXWJVIEFGJ-UHFFFAOYSA-N |
| Vandetanib | ZD6474 | CID 3081361 | UHTHHESEBZOYNR-UHFFFAOYSA-N |
INNs or synonyms are in the first column, the 2nd column is the AZ OI links, 3rd column the PubChem CID, followed by the InChIKey as the 4th column.
UPDATE
13 November 2015
One of fastest development programmes – from start of clinical trials to approval in just over two and a half years to meet unmet patient need
With objective response rate of 59% and duration of response of 12.4 months, TAGRISSO provides important new option for patients
AstraZeneca today announced that the US Food and Drug Administration (FDA) has approved TAGRISSO™ (AZD9291) 80mg once-daily tablets for the treatment of patients with metastatic epidermal growth factor receptor (EGFR) T790M mutation-positive non-small cell lung cancer (NSCLC), as detected by an FDA-approved test, who have progressed on or after EGFR tyrosine kinase inhibitor (TKI) therapy.
AZD9291 is the only approved medicine indicated for patients with metastatic EGFR T790M mutation-positive non-small cell lung cancer. This indication is approved under the FDA’s accelerated approval process based on tumour response rate and duration of response (DoR).
AZD9291 is an EGFR-TKI, a targeted cancer therapy, designed to inhibit both the activating, sensitising mutations (EGFRm), and T790M, a genetic mutation responsible for EGFR-TKI treatment resistance. Nearly two-thirds of NSCLC patients who are EGFR mutation-positive and experience disease progression after being treated with an EGFR-TKI develop the T790M resistance mutation, for which there have been limited treatment options.
Pasi A Jänne MD, PhD, Director, Lowe Center for Thoracic Oncology at Dana-Farber Cancer Institute, Scientific Director, Belfer Center for Applied Cancer Science and Professor of Medicine, Harvard Medical School, said: “In the AURA clinical studies, AZD9291 has demonstrated compelling early efficacy and tolerability in patients with EGFRm T790M metastatic non-small cell lung cancer. This treatment has the potential to become the standard of care for patients living with EGFRm T790M non-small cell lung cancer. The accelerated approval of AZD9291 highlights its clinical promise for a targeted group of patients and gives healthcare providers an important new option.”
Pascal Soriot, Chief Executive Officer, AstraZeneca, said: “The FDA approval of TAGRISSO marks an important milestone for lung cancer patients who urgently need new treatment options. We have built on our heritage in this area and acted on the breakthrough clinical evidence to ensure this next-generation medicine reaches patients in record time. As we advance our comprehensive lung cancer portfolio, we have the opportunity to treat greater numbers of patients across all stages of this disease through precision medicines, immunotherapies and novel combinations.”
AstraZeneca has collaborated with Roche to develop the cobas® EGFR Mutation Test v2 as the companion diagnostic for AZD9291. The cobas® EGFR Mutation Test v2 is intended to identify a range of EGFR mutations in patients with non-small cell lung cancer, including T790M.
AZD9291 was granted Fast Track, Breakthrough Therapy, Priority Review and Accelerated Approval status by the FDA. In Europe and Japan, AZD9291 was granted Accelerated Assessment and Priority Review status respectively. Interactions with regulatory authorities in the rest of the world are ongoing.
The FDA approval of AZD9291 is based on data from the two AURA Phase II studies (AURA extension and AURA2) which demonstrated efficacy in 411 EGFRm T790M NSCLC patients that had progressed on or after an EGFR TKI. In those trials, overall objective response rate ((ORR) a measurement of tumor shrinkage) was 59% (95% CI: 54% to 64%). In a supportive Phase I study in 63 patients, ORR was 51% and median duration of response was 12.4 months.
The AZD9291 tolerability profile showed that no individual severe grade 3+ adverse events occurred at ≥ 3.5%.The most common adverse events were generally mild to moderate and included diarrhoea (42% all grades; 1.0% Grade 3/4), rash (41% all grades; 0.5% Grade 3/4), dry skin (31% all grades; 0% Grade 3/4), and nail toxicity (25% all grades; 0% Grade 3/4). There are no contraindications for AZD9291. Warnings and precautions include interstitial lung disease, QT interval prolongation, cardiomyopathy and embryofoetal toxicity.
AZD9291 Development Programme
AZD9291 is being studied in the confirmatory trial, AURA3, an open label, randomised Phase III study designed to assess the efficacy and safety of AZD9291 versus platinum-based doublet chemotherapy in patients with EGFR T790M positive, locally advanced, or metastatic NSCLC who have progressed following prior therapy with an EGFR-TKI. AZD9291 is also being investigated in the adjuvant setting and in the metastatic first-line setting, including in patients with brain metastases, as well as in combination with other compounds.
NOTES TO EDITORS
About Non-Small Cell Lung Cancer
Lung cancer is the leading cause of cancer death among both men and women, accounting for about one-third of all cancer deaths, more than breast, prostate and colorectal cancers combined. Lung cancer has a five-year survival rate that is less than 20%. Approximately 85% of all lung cancers in the US are NSCLC; 10% to 15% of these are EGFR mutation-positive. Approximately two-thirds of patients treated with EGFR TKI therapy will acquire resistance related to the T790M mutation.
About AZD9291
AZD9291 80mg once-daily tablet is the first medicine indicated for the treatment of patients with metastatic epidermal growth factor receptor (EGFR) T790M mutation-positive non-small-cell lung cancer (NSCLC), as detected by an FDA-approved test, who have progressed on or after EGFR tyrosine kinase inhibitor (TKI) therapy. Non-clinical in vitro studies have demonstrated that AZD9291 has high potency and inhibitory activity against mutant EGFR phosphorylation across the range of clinically relevant EGFRm and T790M mutant NSCLC cell lines with significantly less activity against EGFR in wild-type cell lines.
Osimertinib has recently been published by the World Health Organisation (WHO) as the proposed International Non-proprietary Name (INN) for AZD9291, and may become formally adopted during November 2015. In the US, the American Medical Association accepted osimertinib as the United States Adopted Name (USAN).
About AstraZeneca in Oncology
Oncology is a therapeutic area in which AstraZeneca has deep-rooted heritage. It will be potentially transformational for the company’s future, becoming the sixth growth platform. Our vision is to help patients by redefining the cancer treatment paradigm and one day eliminate cancer as cause of death. By 2020, we are aiming to bring six new cancer medicines to patients.
Our broad pipeline of next-generation medicines is focused on four main disease areas - lung, ovarian, breast, and hematological cancers. These are being targeted through four key platforms – immuno-oncology, the genetic drivers of cancer and resistance, DNA damage repair and antibody drug conjugates.
About Roche
Headquartered in Basel, Switzerland, Roche is a leader in research-focused healthcare with combined strengths in pharmaceuticals and diagnostics. Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and neuroscience. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management. Roche’s personalised healthcare strategy aims at providing medicines and diagnostics that enable tangible improvements in the health, quality of life and survival of patients. Founded in 1896, Roche has been making important contributions to global health for more than a century. Twenty-nine medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and chemotherapy.
About AstraZeneca
AstraZeneca is a global, innovation-driven biopharmaceutical business that focuses on the discovery, development and commercialisation of prescription medicines, primarily for the treatment of cardiovascular, metabolic, respiratory, inflammation, autoimmune, oncology, infection and neuroscience diseases. AstraZeneca operates in over 100 countries and its innovative medicines are used by millions of patients worldwide. For more information please visit www.astrazeneca.com.
CN1C=C(C2=CC=CC=C21)C3=NC(=NC=C3)NC4=C(C=C(C(=C4)NC(=O)C=C)N(C)CCN(C)C)OC