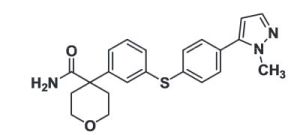
PF 4191834
CAS 1029317-21-2
UNII-YX55DXP4T1; PF-4191834; DVNQWYLVSNPCJZ-UHFFFAOYSA-N;
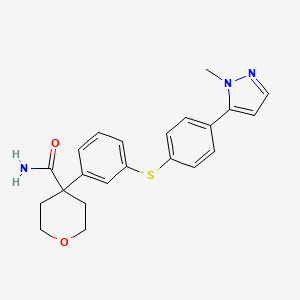
4-(3-{[4-(1-methyl-1H-pyrazol-5-yl)phenyl]thio]phenyl) tetrahydro-2H-pyran-4-carboxamide;
4-[3-[4-(2-methylpyrazol-3-yl)phenyl]sulfanylphenyl]oxane-4-carboxamide
| Molecular Formula: | C22H23N3O2S |
|---|---|
| Molecular Weight: | 393.50192 g/mol |
PF-04191834 works in animal models by inhibiting one of the enzymes, 5-lipoxygenasein which is involved in the pathway that causes inflammation and pain. The purpose of this study is to test how effective, safe and tolerated PF-04191834 is in patients with osteoarthritis of the knee by itself or with naproxen, particularly to test if patients have less pain.
http://www.ncats.nih.gov/files/PF-04191834.pdf
Mechanism:
5-Lipoxygenase (5-LO) inhibitor
Original Development Indication:
AsthmaChronic osteoarthritis pain
PATENT
US 20080125474
formula (Ib):
- Example 1
-
4-(3-bromophenyl)tetrahydro-2H-pyran-4-carbonitrile made by the procedures described in EP 108114 (1.05 kg, 3.95 mole) was stirred in 98% H2SO4 (3.00 L) at room temperature for about 40 h. The mixture was then poured onto ice and the very fine suspension was filtered and washed with H2O thoroughly until pH of wash is neutral. The white solid was washed with hexanes and was then dried in vacuo at 35-40° C. to give 1119 g (99.8% yield) of product in 99.9% purity. LC/MS: 5%-100% CH3CN:H20-0.01% TFA gradient over 10 minutes: 4.68 min. (M+H)+. 1H NMR (400 MHz, DMSO-d6) δ ppm 7.50-7.49 (m, 1H), 7.43-7.40 (m, 1H), 7.36-7.30 (m, 1H), 7.27 (d, J=7.92 Hz, 1H) 7.06 (s, 1H), 5.00 (brs, 1H) 3.71 (dt, J=11.7, 3.7 Hz, 2H), 3.42 (t, J=10.7 Hz, 2H), 2.38 (d, J=13.6 Hz, 2H), 1.75 (td, J=12.2, 4.3 Hz, 2H).
-
Alternative 1
-
4-(3-Bromophenyl)-tetrahydro-2H-pyran-4-carboxamide prepared in step 1 (300 g (1.06 mole), sodium tert-butoxide (122 g, 1.27 mole), Pd(OAc)2 (4.74 g 0.0211 mole) and DIPPF (1,1-bis(diisopropylphosphino)ferrocene) (10.6 g 0.0253 mole) were placed in a flask which was evacuated and filled with N2 3 times. Anhydrous dioxane (2.3 L) was added and the mixture was stirred at room temperature for 1 h. To the mixture was added triisopropylsilane thiol (221 g 1.16 mole) and the resulting mixture was heated to reflux. Reflux was stopped after 1 h and the mixture was allowed to cool to room temperature. The mixture was then poured into ethyl acetate (7 L) which was then washed with H2O (2×4 L) and brine (2 L). The combined aqueous washes were back extracted with ethyl acetate (3 L) which was then washed with H2O (2×2 L) and brine (1 L). The combined organic layers were dried over MgSO4, filtered and concentrated to dryness. Ethyl acetate (0.5 L) was added to the solid and the mixture was stirred on the rotary evaporator to give a fine suspension. Hexanes (1.5 L) was then added and the suspension was allowed to stand for 1 hour. The solid was filtered, washed with 1:1 ethyl acetate-hexanes (1 L) and then hexanes. The resulting brown solid was dried in vacuo to give 334 g (80% yield) of the product in 99% purity. A second crop was obtained from the filtrate which was washed as before and dried to give an additional 15 g product for a total yield of 84%. LC/MS: 5%-100% CH3CN:H20-0.01% TFA gradient over 10 minutes: 9.35 min. 394.1 (M+H)+. 1H NMR (400 MHz, CDCl3) δ ppm 7.52-7.51 (m, 1H) 7.42-7.39 (m, 1H), 7.22-7.21 (m, 2H), 5.35 (brs, 1H), 5.13 (brs, 1H) 3.78-3.75 (m, 4H) 2.36-2.32 (m, 2H), 2.06-2.00 (m, 2H), 1.27-1.16 (m, 3H), 1.05 (d, J=7.25 Hz, 18H).
-
Alternative 2
-
Purge a 3-neck flask (overhead stirrer, nitrogen inlet, serum cap) with nitrogen. Add 4-(3-Bromophenyl)-tetrahydro-2H-pyran-4-carboxamide prepared in step 1 (10 g, 0.03519 mole). Add sodium t-butoxide (4.1 g, 0.04223 moles). Add anhydrous toluene. Toluene should be as dry as possible, <0.01% water by KF is sufficient. Initiate stirring. Purge the reaction mixture with 4 vacuum/nitrogen purge cycles, maintaining 60 torr vacuum for 30 seconds with each cycle. Add the thiol (9.1 g, 0.04223 moles) assuring that oxygen is not introduced into the vessel. Heat to 75° C. Add PdCl2(diphenyl-phosphino ferrocene) (0.258 g, 0.00035 moles). Continue heating to reflux (reaction temperature about 107° C.) for a minimum of 1 hour. The mixture should reach reflux within 30 minutes.
-
Cool the reaction mixture to 25° C. Add ethyl acetate (300 mL, 30 mL/g) and stir the resulting suspension for 30 min. Filter the suspension through celite (30 g). Rinse the celite with ethyl acetate for rinse (100 mL, of product to be rinsed), combining filtrates. Concentrate the filtrate via vacuum distillation at 70 torr at 30° C. until 80% of the filtrate volume has been removed. Add hexane (200 mL, 20 mL/g of product to be crystallized) for crystallization to the slurry over 5 minutes. Stir and cool the mixture to 5° C. Maintain the mixture at 5° C. for a minimum of 1 hour. Isolate product by filtration. Rinse the cake with hexane (100 mL, of product to be rinsed). Dry the cake on the filter to LOD of no more than 5%. Dry the solid at 45-50° C. under vacuum to an LOD of no more than 1.5%. Yield 12 grams (85% yield).
-
Any mL/g amount indicated above is referred to grams of bromo carboxamide.
-
Alternative 1
-
A N,N′-dimethylformamide (15 mL) solution of 4-bromoacetophenone (10.60 g, 53.25 mmols) and N,N′-dimethylformamide dimethyl acetal (2.5 equivalents) was heated at 125 degrees Celcius for 3 hours. The dark red solution was cooled to room temperature. The volatiles were removed by rotary evaporation providing a red viscous oil. To this substance was added anhydrous N,N′-dimethylformamide (15 mL) and methyl hydrazine (7.6 g, 160 mmols, 3 equivalents). The mixture was stirred at room temperature for 1 hour and then heated at 75 degrees Celcius for 4 hours. The volatiles were removed by rotary evaporation and the crude residue was taken up in a small volume of methylene chloride. This red solution was applied to a cartridge of silica gel. The cartridge was eluted with a 20:80 mixture of ethyl acetate and hexanes, respectively. The appropriate fractions were combined and concentrated to produce 12.5 g of a white solid.
-
1H NMR (400 MHz, CHLOROFORM-d) δ ppm 3.87-3.95 (m, J=2.22 Hz, 3H) 6.29-6.36 (m, 1H) 7.31 (dd, J=8.36 Hz, 2H) 7.52-7.56 (m, 1H) 7.62 (dd, J=2.05 Hz, 2H).
-
Alternative 2
-
4-bromoacetophenone (20.0 g; 0.10 mole) and N,N-dimethylformamide dimethylacetal (28.5 mL; 0.20 mole) were mixed together in DMF (12 mL) and heated to 110° C. for 4 hours. The methanol and water that were generated during the reaction were distilled (6.2 mL). The mixture was cooled to 25° C. Methyl t-butyl ether (100 mL) and methylhydrazine (21.2 mL; 0.40 moles) were added and the mixture was stirred over night. The reaction mixture was washed with 1 M aqueous ammonium chloride (3×40 mL) and water (40 mL). The organic phase was dried by azeotropic distillation using a Dean-Stark apparatus. As an alternative to distillation, the solution was dried through an anhydrous magnesium sulfate cartridge. The solution was filtered through a silica gel cartridge (60 g). The product was flushed from the cartridge with methyl t-butyl ether. The fraction(s) containing product were combined and concentrated to about 70 mL by distillation. Heptane (120 mL) was added and distillation was continued until the pot temperature reached 98.4° C. About 100 mL of distillate was collected. The mixture was cooled to 40° C. The mixture was seeded and the temperature was maintained at 40° C. for 30 minutes while crystallization was initiated. The mixture was slowly chilled to 0° C. over 90 minutes. The mixture was held at 0° C. for 30 minutes. The mixture was filtered and the solid was washed (3×) with chilled (0° C.) heptane. The solid was dried on the filter. A cream-colored, crystalline solid (16.3 g; 68% yield) was obtained. The NMR data of the title compound are as per alternative 1.
-
A mixture of 5-(4-bromophenyl)-1-methyl-1H-pyrazole (0.50 g, 2.10 mmols,), 4-{3-[(tri-isopropylsilyl)thio]phenyl}tetrahydro-2H-pyran-4-carboxamide (0.83 g, 2.10 mmols), Tetrakis(triphenylphosphine)palladium(0) (243 mg, 0.10 equivalents), bis[(2-diphenyl-phosphino)]phenyl ether (113 mg, 0.10 equivalents), and 1.0 M potassium tert-butoxide in THF (6.3 mmols, 3 equivalents) in iPrOH (15 mL) that contained 5% water was heated for 4 hours at 90 degrees Celcius in an atmosphere of nitrogen. The reaction mixture was cooled to room temperature and 7 mL of 1N HCl was added. The product was precipitated by the addition of water (30 mL). The precipitate was collected by suction filtration and washed with water (2×20 mL) and cold ethyl ether (4×20 mL). The tan brown solid was dissolved in a small volume of methylene chloride containing 1% methanol and applied to a 140 g cartridge of silica gel. The cartridge was eluted with an acetone:hexane gradient. The appropriate fractions were concentrated and triturated with methanol to produce a white solid (710 mg) as product. 1H NMR (400 MHz, DMSO-d6) δ ppm 1.75-1.84 (m, 3H) 2.40 (d, J=13.54 Hz, 3H) 3.43-3.51 (m, 1H) 3.72 (d, J=11.34 Hz, 3H) 3.84 (s, 3H) 6.40 (d, J=1.46 Hz, 1H) 7.02 (s, 1H) 7.22-7.30 (m, 2H) 7.34 (d, J=8.05 Hz, 1H) 7.38-7.43 (m, 2H) 7.45-7.52 (m, 3H). HRMS calc M+H, 394.1589, found 394.1630.
-
4-{3-[(tri-isopropylsilyl)thio]phenyl}tetrahydro-2H-pyran-4-carboxamide (200 g, 0.51 moles), 5-(4-bromophenyl)-1-methyl-1H-pyrazole (126 g, 0.53 moles), and 2-methyltetrahydrofuran (2,000 mL, 10 mL/g of tips carboxamide) were put into the reactor and sparged with nitrogen while heating to 60° C. The sodium methoxide (244.0 mL, 1.07 moles, added as sodium methoxide in methanol solution 25% w/w) was added to the reactor and sparging was continued for another 30 minutes. PdCl2DPPF (3.7 g, 0.005 moles) was added to the reactor and the mixture was heated to 70° C. Once the amount of tips carboxamide was less than 1% of starting amount, the mixture was cooled to 0° C. The mixture was held at 0° C. for one hour. The mixture was filtered and the solid was washed with 2-methyltetrahydrofuran (3×2.5 mL/g). The solid was dried on the filter. The solid was returned to a clean reactor and triturated with water (2,000 mL, 10 mL/g) for two hours at 20° C. The mixture was filtered and the solid was washed with water (2,000 mL, 2×5 mL/g). The solid was dried on the filter. The solid was returned to a clean reactor with the Si-thiol (90.0 g, 0.5 g/g) and THF (about 12.8 L, 70 mL/g). The mixture was heated to 60-65° C. and held for two hours. The mixture was cooled to 25° C. and filtered. The Si-thiol was washed with THF (about 0.9 L, 5 mL/g). The solution was distilled to a concentration of 10 mL/g. The mixture was cooled to 25° C. and hexanes (422.5 mL, 5 mL/g) was added. The mixture was filtered and the solid was washed with hexanes (422.5 mL, 5 mL/g). The solid was dried in a vacuum oven at 70° C.
-
For 2-methyltetrahydrofuran and water, mL/g are referred to grams of tips carboxamide. For Si-thiol, tetrahydrofuran and hexanes, mL/g are referred to grams of title compound.
PAPER
Transition Metal-Catalyzed Couplings in Process Chemistry (2013), 253-266
Book Title
Transition Metal-Catalyzed Couplings in Process Chemistry: Case Studies from the Pharmaceutical Industry
18. Development of Migita Couplings for the Manufacture of a 5-Lipoxygenase Inhibitor
Published Online: 19 JUL 2013
DOI: 10.1002/9783527658909.ch18
- 5-lipoxygenase inhibitor;
- isooctyl 3-mercaptopropionate;
- Migita couplings;
- one-pot process;
- triisopropylsilanethiol (TIPS-SH)
Summary
The biggest shortcoming of the medicinal chemistry route is the introduction of the sulfur source for the first of two Migita couplings. The authors felt that the initial Migita coupling was a better candidate for a kinetic study on the formation of impurity, as it was harder to maintain a constant concentration of active Pd for the second coupling with two sources of Pd/ligand in this step. As depicted in the mechanism of the Migita coupling, the catalytic cycle is composed of three steps: oxidative addition, transmetalation, and reductive elimination. This chapter develops a three-step, one-pot process for the synthesis of 5-lipoxygenase inhibitor via a sequence of two Migita couplings. This strategy employed cheap, odorless, and readily available isooctyl 3-mercaptopropionate as the sulfur source for the initial Migita coupling as a general alternative to the popular triisopropylsilanethiol (TIPS-SH) for the formation of diaryl thioethers.
PAPER
PAPER
Brian Chekal, David Damon, Danny LaFrance, Kyle Leeman, Carlos Mojica, Andrew Palm, Michael St. Pierre, Janice Sieser, Karen Sutherland, Rajappa Vaidyanathan, John Van Alsten, Brian Vanderplas, Carrie Wager, Gerald Weisenburger, Gregory Withbroe, and Shu Yu
Publication Date (Web): July 17, 2015 (Article)
DOI: 10.1021/op500412a
A de novo three-step-one-pot process for the formation of PF-04191834 was developed. This methodology employed inexpensive, odorless, and readily available commodity chemical iso-octyl-3-mercaptopropionate as a sulfur source, which could be a general alternative to the popular TIPS-SH in the formation of diarylthioethers via Migita coupling. A kinetic study revealed that, at high temperature, reductive elimination could be the rate-limiting step in the catalytic cycle, which opens pathways for the generation of undesired impurities. By proper control of the reaction conditions, the desired API was synthesized in >70% crude yield and in 55% isolated yield after vigorous purifications. This process was successfully demonstrated on a 20 kg scale.
Pure API after drying under vacuum. Mp 173 °C.
1H NMR (400 MHz, DMSO-d6) 7.52 (2H, m), 7.48 (2H, m), 7.42 (2H, m), 7.35 (2H, m), 7.29 (2H, m), 7.07 (1H, br. s), 6.42 (1H, d, J = 1.8 Hz), 3.85 (3H, s), 3.74 (2H, dt, J = 11.7, 3.7 Hz), 3.47 (2H, br. t, J = 11.7 Hz), 2.41 (2H, br. d, J = 13.3 Hz), 1.80 (2H, m).
13C NMR (100.6 MHz, DMSO-d6) 174.6, 146.0, 141.9, 137.9, 136.0, 133.2, 130.1, 129.7, 129.4, 129.3, 128.6, 125.6, 105.9, 64.6, 47.8, 37.6, 33.9.
LCMS: found m/z 394.17 [M + H]+. Anal. Calcd for C22H23N3O2S: C, 67.15; H, 5.89; N, 10.68; S, 8.15. Found: C, 67.09; H, 5.93; N, 10.69; S, 8.16.
After pd removal
Mp 173 °C.
1H NMR (400 MHz, DMSO-d6) 7.52 (2H, m), 7.48 (2H, m), 7.42 (2H, m), 7.35 (2H, m), 7.29 (2H, m), 7.07 (1H, br. s), 6.42 (1H, d, J = 1.8 Hz), 3.85 (3H, s), 3.74 (2H, dt, J = 11.7, 3.7 Hz), 3.47 (2H, br. t, J = 11.7 Hz), 2.41 (2H, br. d, J = 13.3 Hz), 1.80 (2H, m).
13C NMR (100.6 MHz, DMSO-d6) 174.6, 146.0, 141.9, 137.9, 136.0, 133.2, 130.1, 129.7, 129.4, 129.3, 128.6, 125.6, 105.9, 64.6, 47.8, 37.6, 33.9.
LCMS: found m/z 394.17 [M + H]+. Anal. Calcd for C22H23N3O2S: C, 67.15; H, 5.89; N, 10.68; S, 8.15. Found: C, 67.09; H, 5.93; N, 10.69; S, 8.16.
| Patent | Submitted | Granted |
|---|---|---|
| Pyrazole Analogs [US7772269] | 2008-05-29 | 2010-08-10 |
| Pyrazole Derivatives as 5-LO-Inhibitors [US8097733] | 2009-09-10 | 2012-01-17 |
| NOVEL TREATMENT FOR AGE RELATED MACULAR DEGENERATION AND OCULAR ISCHEMIC DISEASE ASSOCIATED WITH COMPLEMENT ACTIVATION BY TARGETING 5-LIPOXYGENASE [US2011269807] | 2011-11-03 | |
| TREATMENT AND PREVENTION OF DISEASES MEDIATED BY MICROORGANISMS VIA DRUG-MEDIATED MANIPULATION OF THE EICOSANOID BALANCE [US2014171445] | 2012-08-02 | 2014-06-19 |
SEE........http://newdrugapprovals.org/2015/12/14/pf-04191834-for-patients-with-osteoarthritis-of-the-knee/
////////
c1c(cc(cc1)C2(C(=O)N)CCOCC2)Sc3ccc(cc3)c4ccnn4C or
CN1C(=CC=N1)C2=CC=C(C=C2)SC3=CC=CC(=C3)C4(CCOCC4)C(=O)N